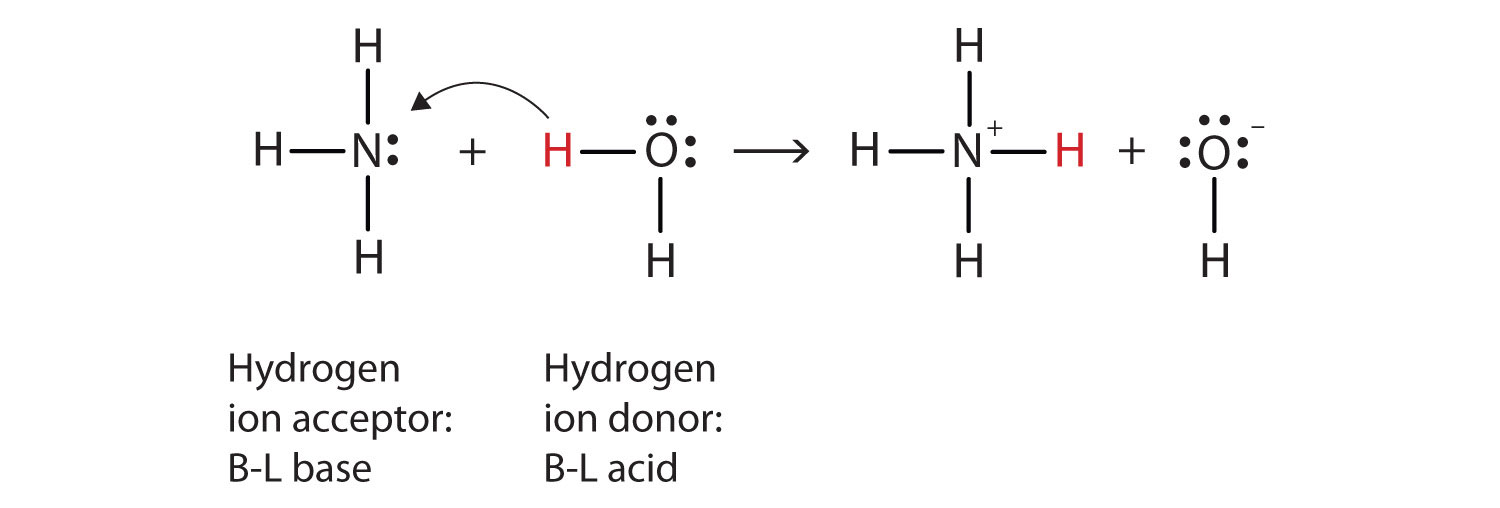
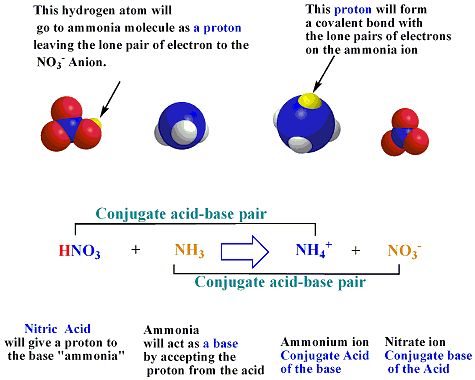
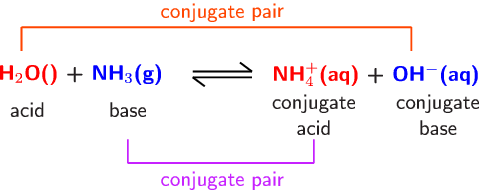
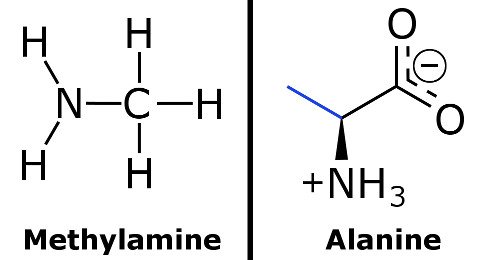
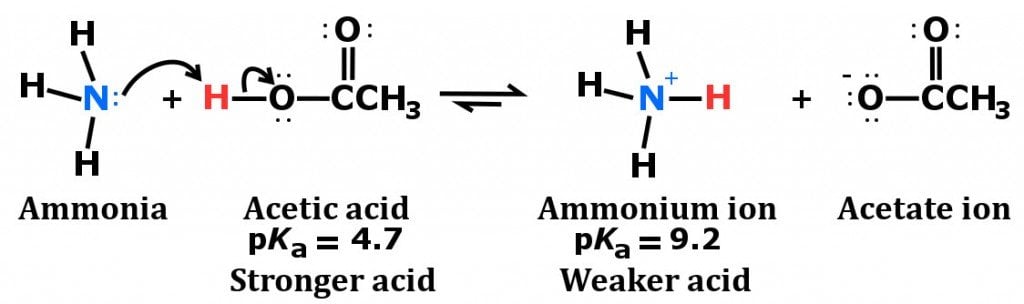
Spontaneity of the acid–base reaction between acetic acid and ammonia... | Download Scientific Diagram

Highly efficient and selective separation of ammonia by deep eutectic solvents through cooperative acid-base and strong hydrogen-bond interaction - ScienceDirect

Question Video: Identifying the Lewis Acid in the Reaction of Ammonia with Boron Trifluoride | Nagwa

a) Explain why ammonia acts as a weak base in water. (b) Write a balanced chemical equation for the reaction between ammonia and water. | Homework.Study.com
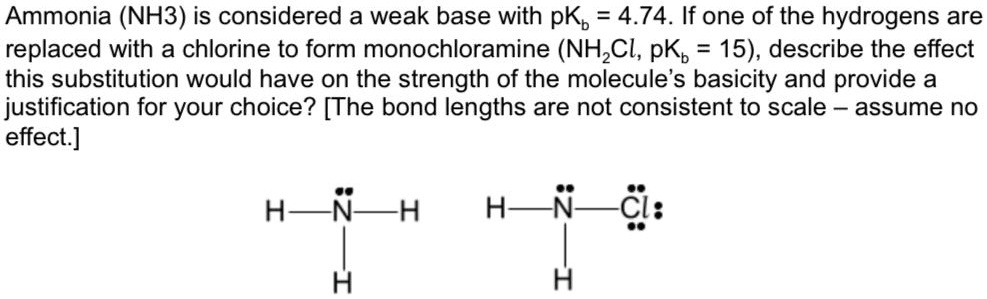
SOLVED: Ammonia (NH3) is considered a weak base with pKb = 4.74. If one of the hydrogens are replaced with a chlorine to form monochloramine (NHZCl, pKb 15) , describe the effect

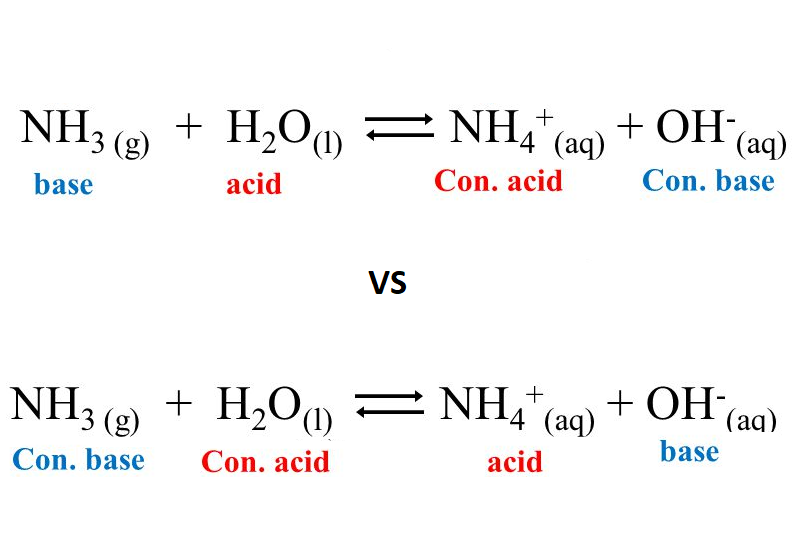
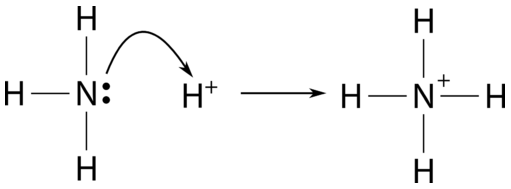




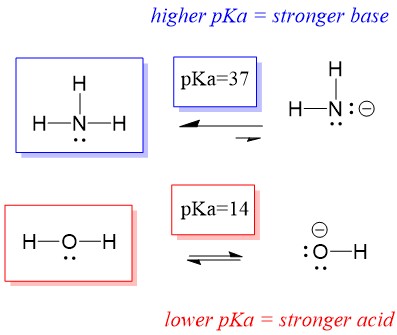
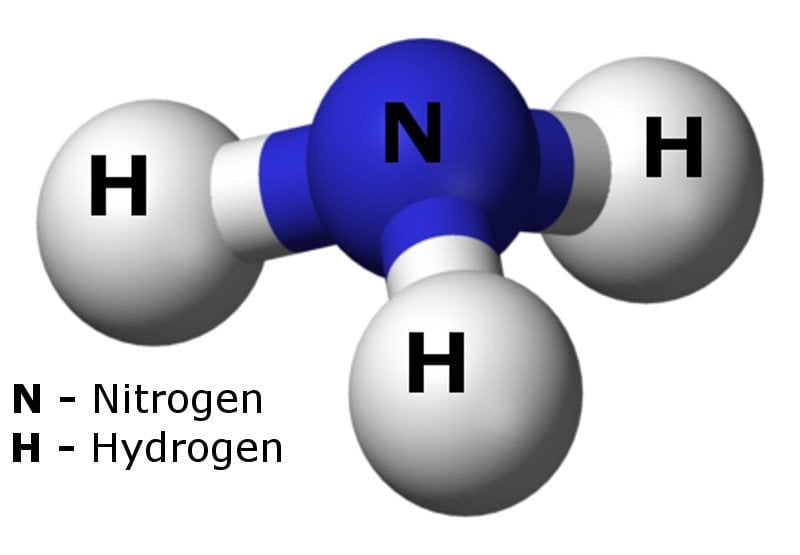
![Why does \\[N{H_3}\\] act as a Lewis base? Why does \\[N{H_3}\\] act as a Lewis base?](https://www.vedantu.com/question-sets/db470ea0-7477-412c-82d8-a4917a1132145281377342200076057.png)